Overview
Our lab studies how cells detect and degrade aberrant RNAs, and what happens when this surveillance fails. We combine computational biology, functional genomics, and biochemistry to uncover the rules governing RNA quality control, with implications for genetic disease diagnosis and therapy.
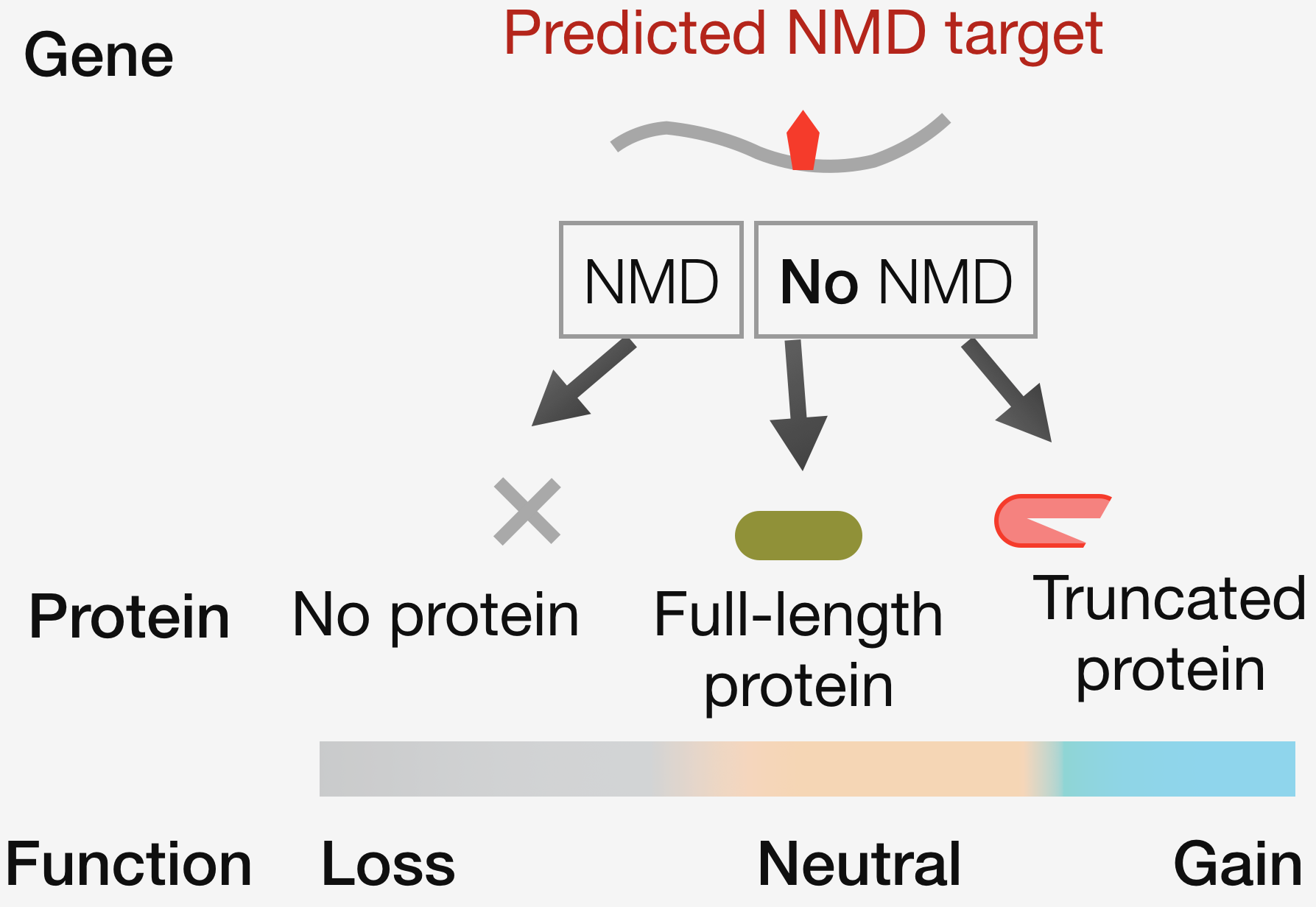
Robustness in biological systems relies on quality control. While errors in the DNA are repaired to maintain fidelity in the flow of genetic information, errors in mRNA are dealt with by degrading the erroneous molecules via surveillance processes such as nonsense-mediated mRNA decay (NMD). Though all mRNAs degraded by NMD follow a set of canonical rules, the converse – that every mRNA that follows the rule is subject to NMD – is not true. This variation in RNA quality control is a major confounding factor in interpreting the clinical impact of human genetic variants. An NMD target may degrade and generate no protein, or escape degradation and result in the production of a full-length or truncated protein – each scenario associated with a potentially different outcome for the cell. The long-term goal of our research program is to understand the molecular logic governing variation in RNA quality control, and to translate these insights into improved variant interpretation and therapies for genetic diseases.
Our questions
- What cis elements and trans factors allow an mRNA to escape NMD?
- How does variable NMD sensitivity influence cellular phenotypes?
- What is the role of loss of NMD in facioscapulohumeral muscular dystrophy (FSHD)?
- How can we leverage these insights to improve clinical variant interpretation and develop therapies for rare diseases?
Our Systems and tools
We study RNA surveillance across biological scales, from individual transcripts to population-level analyses. Our experimental systems include human cell lines, patient-derived fibroblasts, and primary muscle cells, allowing us to connect molecular mechanisms to disease-relevant contexts. We also mine large-scale human genetic databases to understand how RNA quality control shapes the landscape of genetic variation.
Our methods span computational and experimental approaches: massively parallel reporter assays to test thousands of sequence variants simultaneously, saturation genome editing to assess variant effects at endogenous loci, ribosome profiling and quantitative proteomics to measure protein output, and targeted biochemistry to dissect mechanism. We develop and share tools, including reporter systems and computational pipelines, to enable the broader community to study RNA surveillance.